December 3rd, 2011 by Elaine Schattner, M.D. in Opinion, Research
No Comments »

The latest issue of the Annals of Internal Medicine contains 2 noteworthy papers on cervical cancer screening. The first, a systematic review of studies commissioned by the USPSTF, looked at 3 methods for evaluating abnormalities in women over 30 years:
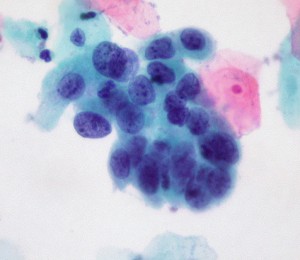
high-grade cervical cell dysplasia (Dr. E. Uthman, Wikimedia Commons)
1. Conventional cytology (as in a Pap smear; the cervix is scraped and cells splayed onto a microscope slide for examination);
2. Liquid-based cytology (for LBC, the NHS explains: the sample is taken as for a Pap test, but the tip of the collection spatula is inserted into fluid rather than applied to slides. The fluid is sent to the path lab for analysis);
3. Testing for high-risk HPV (human papillomavirus). Currently 3 tests have been approved by the FDA in women with atypical cervical cells or for cervical cancer risk assessment in women over the age of 30: Digene Hybrid Capture 2 (manufactured by Quiagen), Cobas 4800 HPV (Roche) and Cervista HR HPV (Hologic); another Roche Diagnostics assay, Amplicor HPV, awaits approval.
These HPV assays use distinct methods to assess DNA of various HPV strains.
There’s a lot of jargon here, and I have to admit some of this was new to me despite my nearly-due diligence as a patient at the gynecologist’s office and my familiarity as an oncologist with the staging, clinical manifestations and treatment of cervical cancer. Who knew so many decisions were made during a routine pelvic exam about which manner of screening? Read more »
*This blog post was originally published at Medical Lessons*
October 12th, 2011 by Peggy Polaneczky, M.D. in Health Tips, Opinion
No Comments »

 A well-written and balanced article on mammography from USA Today may help move the conversation about this screening test away from hype and a bit closer to reality. The title – “Mammogram is ‘terribly imperfect’, though recommended.”
A well-written and balanced article on mammography from USA Today may help move the conversation about this screening test away from hype and a bit closer to reality. The title – “Mammogram is ‘terribly imperfect’, though recommended.”
For women in their 40s, mammograms reduce the risk of dying from breast cancer by about 15%… But mammograms miss some cancers and raise false alarms about others, causing women to go through unnecessary follow-up tests… “We’re saying, ‘Mammography is a terribly imperfect test, but we’re recommending women get it,’” Brawley says. “The task force was saying, ‘Mammography is a terribly imperfect test, and Read more »
*This blog post was originally published at The Blog That Ate Manhattan*
October 1st, 2011 by KennyLinMD in Book Reviews, Opinion
No Comments »

 Recently, I had a conversation with Shannon Brownlee (the widely respected science journalist and acting director of the Health Policy Program at the New America Foundation) about whether men should continue to have access to the PSA test for prostate cancer screening, despite the overwhelming evidence that it extends few, if any, lives and harms many more men than it benefits. She felt that if patients could be provided with truly unbiased information and appropriate decision aids, they should still be able to choose to have the test (and have it covered by medical insurance). Believing that one of the most important roles of doctors is to prevent patients from making bad decisions, I disagreed.
Recently, I had a conversation with Shannon Brownlee (the widely respected science journalist and acting director of the Health Policy Program at the New America Foundation) about whether men should continue to have access to the PSA test for prostate cancer screening, despite the overwhelming evidence that it extends few, if any, lives and harms many more men than it benefits. She felt that if patients could be provided with truly unbiased information and appropriate decision aids, they should still be able to choose to have the test (and have it covered by medical insurance). Believing that one of the most important roles of doctors is to prevent patients from making bad decisions, I disagreed.
After reading Your Medical Mind, the new book by Harvard oncologist and New Yorker columnist Jerome Groopman, I think he would probably side with Brownlee’s point of view. Groopman, whose authoring credits include the 2007 bestseller How Doctors Think, and wife Pamela Hartzband, MD have written a kind of sequel to that book that could have easily been titled How Patients Think. Drawing on interviews with dozens of patients about a wide variety of medical decisions – from starting a cholesterol-lowering drug, to having knee surgery, to accepting or refusing heroic end-of-life interventions – the authors Read more »
*This blog post was originally published at Common Sense Family Doctor*
September 25th, 2011 by GarySchwitzer in Opinion
No Comments »

In a highly-promoted appearance, legendary Florida State football coach Bobby Bowden went on ABC’s Good Morning America yesterday to announce that he had kept silent since 2007 about his diagnosis with prostate cancer.
First, let me say that I’ve always liked this guy. Funny. Charming. Coached teams that were fun to watch.
But that doesn’t make you an effective communicator on prostate cancer.
If you listen very carefully to the following clip (it took me 3 times watching the clip before I caught this), you’ll hear interviewer Robin Roberts rapidly mention that Bowden “is being compensated” for his appearance by “On the Line.”
“On the Line” is sponsored by several entities including two drug companies that make prostate cancer drugs and by Project Zero – whose executive made news on this blog recently by writing that Dr. Otis Brawley of the American Cancer Society “has killed more men by giving them an excuse to not be tested.”
You could probably find less conflicted sources Read more »
*This blog post was originally published at Gary Schwitzer's HealthNewsReview Blog*
July 7th, 2011 by admin in Research
No Comments »

Nearly forty years ago, President Richard Nixon famously declared a “War on Cancer” by signing the National Cancer Act of 1971. Like the Manhattan Project, the Apollo program that was then landing men on the Moon, and the ongoing (and eventually successful) World Health Organization-led initiative to eradicate smallpox from the face of the Earth, the “War on Cancer” was envisioned as a massive, all-out research and treatment effort. We would bomb cancer into submission with powerful regimens of chemotherapy, experts promised, or, failing that, we would invest in early detection of cancers so that they could be more easily cured at earlier stages.
It was in the spirit of the latter that the National Cancer Institute launched the Prostate, Lung, Colorectal, and Ovarian Cancer (PLCO) Screening trial in 1992. This massive study, which eventually enrolled more than 150,000 men and women between age 55 and 74, was designed to test the widespread belief that screening and early detection of the most common cancers could improve morbidity and mortality in the long term. Not a few influential voices suggested that the many millions of dollars invested in running the trial might be better spent on programs to increase the use of these obviously-effective tests in clinical practice.
They were wrong. As of now, the PLCO study is 0-for-2. Read more »
*This blog post was originally published at Prepared Patient Forum: What It Takes Blog*




















